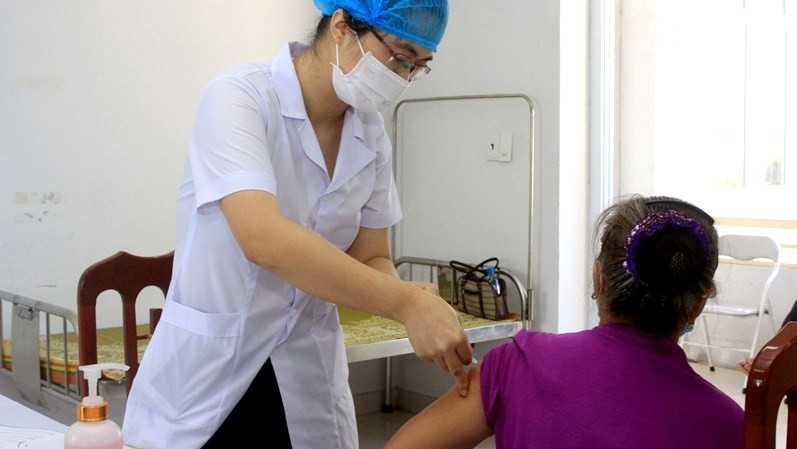
The northern province is the only locality nationwide selected for the second phase trials. About 80 volunteers are expected to receive the first shot of the vaccine on August 18.
Covivac has been developed and produced by the Institute of Vaccines and Medical Biologicals (IVAC). It is the second homegrown vaccine approved by the Ministry of Health for clinical trials, after Nanocovax. The vaccine has been given to 100 volunteers at Hanoi Medical University during the first phase of human trials.
In the second phase, a total of 375 volunteers, aged over 18, have been chosen and classified into three groups for the 3 mcg and 6 mcg dosages and a dose of AstraZeneca vaccine.
According to IVAC Director Duong Huu Thai, AstraZeneca vaccine is being used in place of a placebo, which would compare the immunogenicity of the two vaccines. Blood samples collected from the second phase will be sent to Canada for an independent evaluation.
The research team expects to finish the injection of the first jabs on August 23 and the second shots will be administered to the volunteers after 28 days.